- Log in to post comments
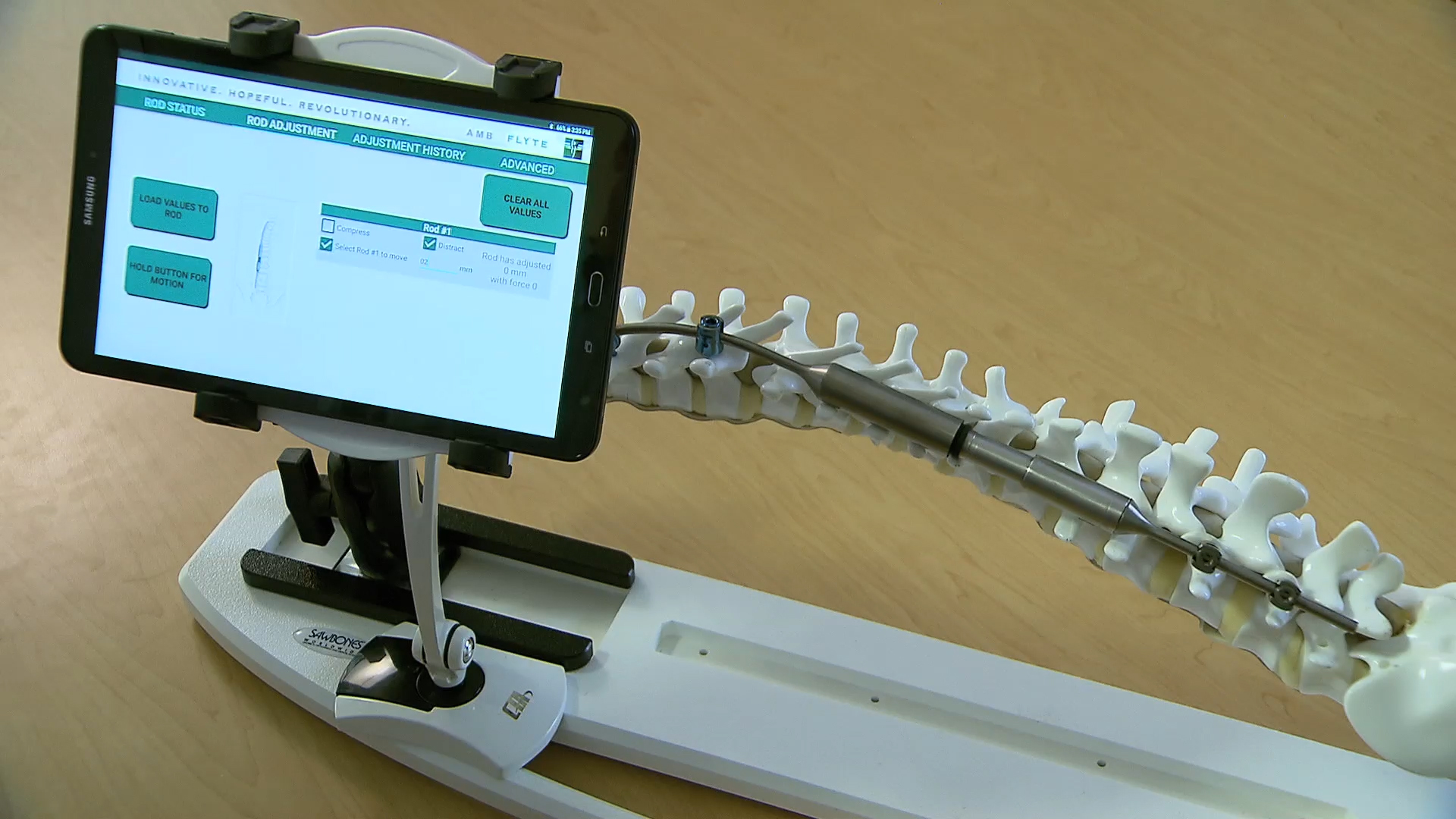
DAYTON—With a functional prototype complete, AMB Surgical now moves to miniaturize their patented FLYTE™ Smart Automated Growing Rod, a medical device that could revolutionize the way physicians treat juvenile scoliosis, lengthen limbs, and stabilize traumatic injuries.
“From the very beginning, we believed our concept would work,” said AMB founder Casel Burnett. “We’ve now shown our software-controlled growth rod can adjust using our technology. Establishing this ‘proof of concept’ validates our years of work and positions us to move forward with reducing the size of the device to be used on smaller patients.”
The Dayton start-up uses patented technology to non-surgically automate and remotely control the extension of rods used in orthopedic surgeries. Current devices use manual adjustments, either by having caretakers, such as parents, use tools to adjust an external framework for limb extension, or, for scoliosis, through repeated back surgeries. A physician implants an adjustable rod along the child’s spine, and as the child grows, uses tools to lengthen the rod through additional surgeries to correct the curve.
The key to AMB’s technology is an innovative combination of miniature gearing, power, sensors and wireless communication technology located inside the rod. The rod is controlled remotely by a software application that programs the rod, makes surgeon-programmed adjustments, records and monitors real-time biomechanical information over the course of treatment. The technology has the promise to significantly reduce the number of surgeries and cost while improving patient outcomes.
AMB began working with the Center for Design and Manufacturing Excellence (CDME) at The Ohio State University earlier this year to produce a Phase 1 functional prototype and complete successful testing.
“We were able to take the concept for AMB’s FLYTE through the design process to a working prototype that demonstrates key functionality critical to advancing the product to the next phase of development,” said Mary Hoffman Pancake, Program Manager for Biomedical Devices at CDME. “The CDME-AMB collaboration meshed perfectly with our mission to meet the translational research needs of university partners by using applied engineering to help commercialize innovations that provide a direct impact on society.”
The product’s next phase will focus on readying the technology for clinical testing and approval from the Federal Drug Administration. It’s an exciting time for a company driven by personal mission to make scoliosis treatment less traumatic for young patients.
Burnett’s daughter was diagnosed with juvenile scoliosis as a young child and endured years of invasive surgeries as part of her treatment. Scoliosis, or curvature of the spine, can lead to life-long health issues if not corrected while the child is still growing. Watching his daughter undergo treatment motivated him to find a better way.
Burnett is a mechanical engineer and group manager of production engineering at Toyota. He partnered with his friend, Tyson Ross, an electrical controls engineer and program manager at the U.S. Air Force Research Laboratory, to see how they could take the existing technology and improve it.
FLYTE would expand the rod remotely, without surgery, as the child grows. The significance of this unique feature is that it reduces the number of surgeries and potentially accelerates the treatment. Furthermore, this mechanism would also eliminate the need for parents to adjust external frames, reducing the likelihood of missing adjustments.
For physicians, the device would also provide valuable feedback they can’t access today. The same technology that allows the device to adjust, allows it to collect data on the patient’s progress. The company already holds patents on the technology, and the initial research and design have generated interest from world leaders in pediatric orthopaedic surgery and leading orthopaedic and spine implant companies.
Dr. Michael Albert, Director of Orthopaedics at Dayton Children’s Hospital, called the product a “game changer” in this type of treatment. On Sept. 28, Dr. Albert, an advisor to AMB, presented the device to an orthopaedic treatment symposium at Dayton Children’s Hospital.
In the coming months, AMB plans to collaborate with Battelle to further develop and test the device in preparation for regulatory approval. Battelle's vision is to be a major force in science and technology discovery and in the translation of knowledge into innovative applications that have significant societal and economic impacts.
“Battelle is a recognized leader in developing novel technologies for medical applications with extensive engineering and testing capabilities and is ideally suited for AMB Surgical’s initiatives at this time,”said Dan Sands, CEO of AMB Surgical.
Company leaders will be participating in several presentations in the coming weeks, sharing their progress with the medical and investor community.
Interested in learning more? Meet the AMB team at the following events:
Oct 2nd and 3rd - The Medical Alley Innovation Summit Event in Minneapolis, MN
Oct 16th – The Ohio Defense Forum 2018 – Columbus, OH
Oct 23rd - Musculoskeletal New Ventures Conference – Memphis, TN
About AMB Surgical II, LLC:
AMB Surgical is devoted to the development and commercialization of its FLYTE™ smart automated growing rod technology which holds promise to reduce anxiety and trauma from juvenile and adolescent surgeries. The invasive nature of surgery is, in itself, a difficult reality and one that impedes an individual’s ability to comfortably enjoy life. AMB Surgical’s groundbreaking, patented technology is intended to reduce the necessity for invasive and repetitive surgery in children and teens with orthopedic deformities such as scoliosis and limb abnormalities. FLYTE is a sophisticated electro-mechanical, software activated device to provide real-time biomechanical feedback to guide treatment, as well as, allow surgeons to perform accurate, non-surgical skeletal adjustments. Through our work, AMB Surgical is advancing the boundaries of modern medicinal practices.
Learn more about AMB Surgical at www.ambsurgical.com, or contact Dan Sands at dan.sands@AMBsurgical.com.
Learn more about the Center for Design and Manufacturing Excellence (CDME) at The Ohio State University at https://cdme.osu.edu/about-cdme-0 or contact Elizabeth Culley at culley.36@osu.edu.